- Posts: 21
- Thank you received: 13
 5% HCL
5% HCL
- Mikey99
-
Topic Author
- Offline
- Junior Member
-

Less
More
14 Mar 2020 15:35 #62947
by Mikey99
5% HCL was created by Mikey99
I know this question has been raised before but I cannot seem to find the link I can purchase 5% HCL at a reasonable cost and would like to know if this would substitute for the 4% recommended Also if I were to dilute it what would be the amount of water I would need to add to 100ml acid at 5%
Thanks in advance
Thanks in advance
Please Log in or Create an account to join the conversation.
- mart1n
-

- Offline
- Platinum Member
-
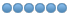
Less
More
- Posts: 450
- Thank you received: 385
14 Mar 2020 19:53 - 14 Mar 2020 20:14 #62953
by mart1n
Replied by mart1n on topic 5% HCL
Hi Mikey
2 images/illustrations below on how to use the endemo calculator
Instead of 30% stock solution you will be using 5% stock solution. Fill in the amount (volume) of your stock solution (in ml) as well and calculate.
I get my 30% HCL at hardware stores. You want clear HCL. No added color. If in doubt ask for a datasheet. Should be HCL dissolved in water, nothing else.
Always add acid to water. Good luck.
2 images/illustrations below on how to use the endemo calculator
Instead of 30% stock solution you will be using 5% stock solution. Fill in the amount (volume) of your stock solution (in ml) as well and calculate.
I get my 30% HCL at hardware stores. You want clear HCL. No added color. If in doubt ask for a datasheet. Should be HCL dissolved in water, nothing else.
Always add acid to water. Good luck.
Last edit: 14 Mar 2020 20:14 by mart1n.
The following user(s) said Thank You: CLO2, Mikey99
Please Log in or Create an account to join the conversation.
- Mikey99
-
Topic Author
- Offline
- Junior Member
-

Less
More
- Posts: 21
- Thank you received: 13
15 Mar 2020 13:07 #62957
by Mikey99
Replied by Mikey99 on topic 5% HCL
Thanks for the link
So for 5% HCL in 100 ml the calculator gives me 125 ml @ 4% As there must be 5 ml of HCL included in the solution then I assume need to add 20 ml
Is this correct
Thanks
So for 5% HCL in 100 ml the calculator gives me 125 ml @ 4% As there must be 5 ml of HCL included in the solution then I assume need to add 20 ml
Is this correct
Thanks
Please Log in or Create an account to join the conversation.
- CLO2
-

- Offline
- Administrator
-

15 Mar 2020 19:28 - 15 Mar 2020 19:35 #62964
by CLO2
Replied by CLO2 on topic 5% HCL
Last edit: 15 Mar 2020 19:35 by CLO2.
The following user(s) said Thank You: Mikey99, JamesC
Please Log in or Create an account to join the conversation.
Time to create page: 0.320 seconds